Rare diseases should be treated free of charge.
Date:2019/7/3
Review and Approval Status of the First Batch of Overseas New Drugs with Urgent Clinical Needs by CDE (Note: Some drugs have not been updated to their current progress; statistics may be current as of December 2018?)
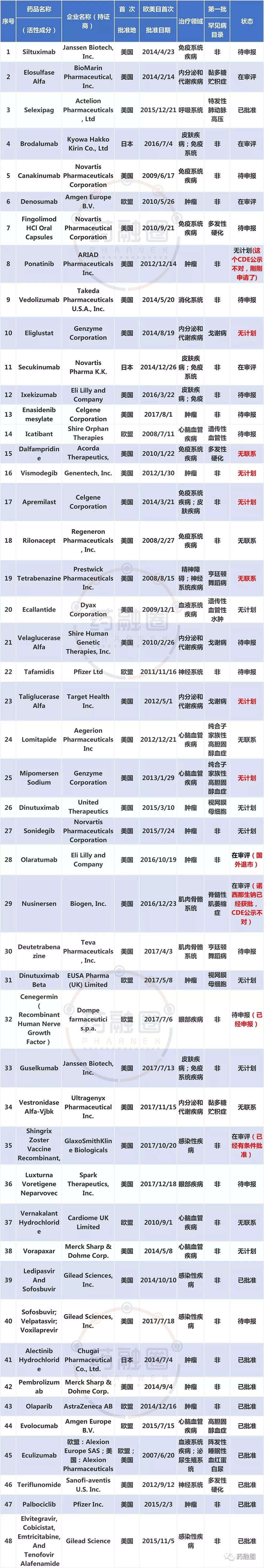
In recent years, foreign pharmaceutical companies have continuously entered China's rare disease market. To encourage domestic enterprises to increase R&D investment in rare disease drugs, the Chinese government has been introducing policies to provide incentives and guidance. In December 2017, the former China Food and Drug Administration (CFDA) issued the “Opinion on Encouraging Drug Innovation and Implementing Priority Review and Approval,” which included rare disease treatments within the scope of priority review. On May 11, 2018, five ministries—the National Health Commission, Ministry of Science and Technology, Ministry of Industry and Information Technology, National Medical Products Administration, and National Administration of Traditional Chinese Medicine—jointly released the “First Batch of Rare Disease Catalog.” This national rare disease list, encompassing 121 conditions, marked China's first official definition of rare diseases. On February 11, 2019, Premier Li Keqiang presided over a State Council executive meeting, which emphasized ensuring access to medications for over 20 million rare disease patients. On February 15, 2019, the National Health Commission's official website released the “Notice of the General Office of the National Health Commission on Establishing a National Rare Disease Diagnosis and Treatment Collaborative Network” (hereinafter referred to as the “Notice”). This initiative selected 324 hospitals with strong capabilities and extensive experience in diagnosing and treating rare diseases to form a collaborative network.
However, these policies have yielded limited results. Few domestic companies were originally involved in orphan drug development. Although policies provide support and guidance, tangible benefits remain scarce. Some investors may prefer early-stage funding, while later financing rounds face challenges due to clinical trial risks, payment mechanisms, and other considerations.
Rare diseases refer to conditions with extremely low incidence rates. Also known as “orphan diseases,” they lack a clear definition in China. According to the World Health Organization (WHO), a rare disease affects between 0.65‰ and 1‰ of the total population. Countries worldwide have varying criteria for identifying rare diseases based on their specific circumstances. For instance, the United States defines rare diseases as those affecting fewer than 200,000 people annually (or less than 1 in 1,500 of the population). Japan specifies rare diseases as those affecting fewer than 50,000 people (or 1 in 2,500 of the population), while Taiwan uses an incidence rate of less than 1 in 10,000 as the criterion.
In early 2018, the “2018 China Rare Disease Research Report,” jointly released by the Beijing Pain Challenge Foundation and Hong Kong Baptist University, revealed that 33.3% of physicians admitted they had only heard of rare diseases but lacked detailed knowledge; 1.1% had never even heard of them.
Yu Shihui, a clinical geneticist at the American College of Medical Genetics and Genomics and Chief Scientific Officer at KingMed Diagnostics, candidly noted that while rare diseases encompass numerous conditions, their low prevalence makes diagnosis exceptionally challenging. Data indicates that a rare disease patient typically consults 5 to 10 physicians, with diagnosis taking 5 to 30 years and a misdiagnosis rate nearing 50%. Moreover, rare diseases are often severe and even life-threatening, imposing heavy financial and emotional burdens on affected families.
Given this, we believe the government should establish a system to create a rare disease fund, providing free treatment for patients. Such a policy could potentially stimulate domestic research into rare disease medications, incentivizing companies to explore this field.



