Pharmaceutical Online has released new guidelines on cleaning validation—Cleaning Validation for the 21st Century. The 100-page document comprises the following eight sections:

Developing Clean Processes and Validation Based on Scientific, Risk, and Statistical Methods
Using ADE to Assess Cross-Contamination Risks in Shared Facility Products
Process Capability Approach for Evaluating Cross-Contamination Risks in Shared Facilities
Swab Method for Assessing TOC Detectability
Validating Visual Inspection as an Analytical Method for Cleaning Validation
Visual Inspection for Justification and Confirmation in Low-Risk, Multi-Product Shared Facility Cleaning Validation
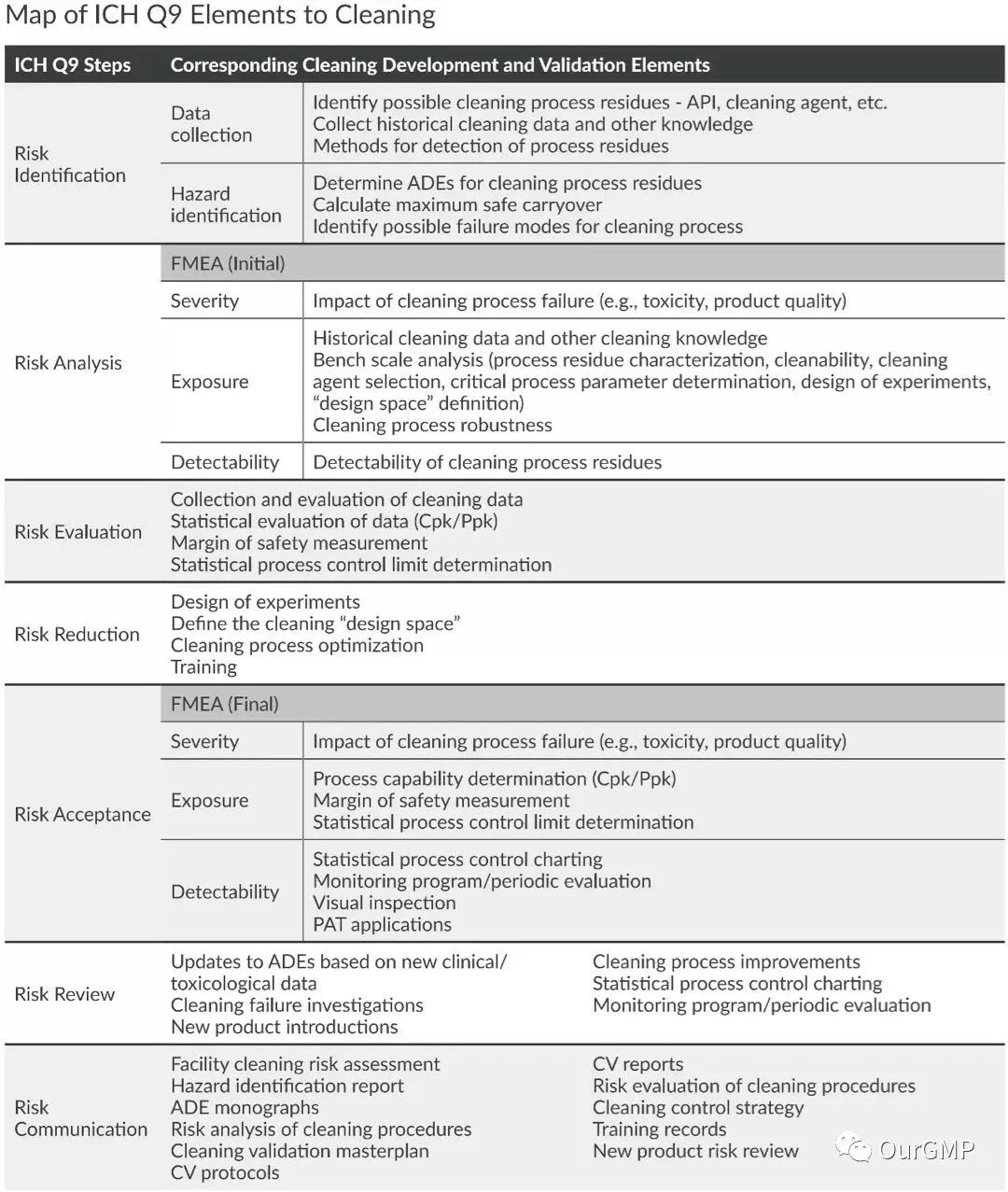

Guidance on the application of the FDA Guidance for Industry: Process Validation—General Principles and Requirements to the development and validation of cleaning processes:
For scoring cross-contamination risks, a scoring system based on ADE values is provided. For example, an ADE of 1 g/day corresponds to a risk score of 0; conversely, an ADE of 100 pg/day corresponds to a risk score of 10.

The article provides an example where four workshops—A, B, C, and D—each produce five products and have different ADE risk scores:
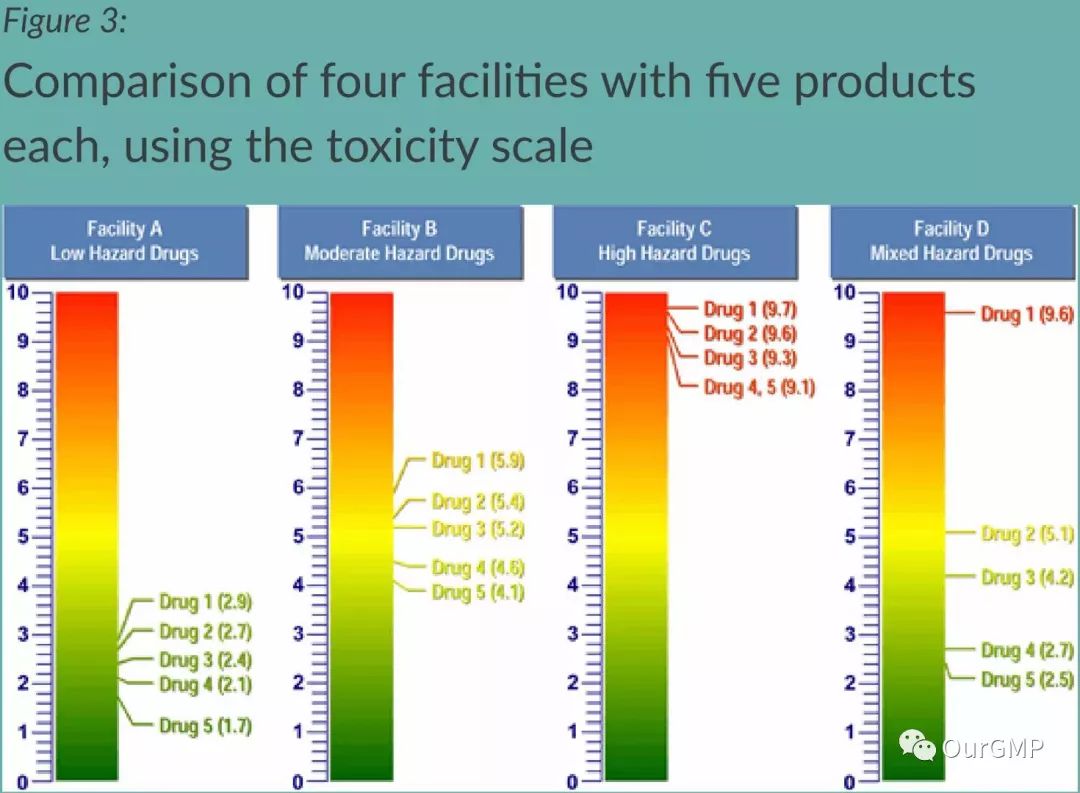
For validation strategies, Workshop A may not require the same validation procedures as Workshop C. If the calculated Maximum Surface Residue (MSSR) is significantly above visible levels and all surfaces can be inspected, “visual inspection” may serve as the sole acceptable criterion for its cleaning validation. Conversely, Workshop C's MSSR may fall below visible levels, necessitating wipe/rinse sampling and potentially requiring specific analytical methods, possibly with ongoing monitoring. Workshop D handles low-, medium-, and high-toxicity drugs on the same line. In this scenario, comprehensive production controls, swabbing/rinsing, sampling, and continuous monitoring may be required after drug 1 (toxicity score = 9.6). However, drugs 4 (toxicity score = 2.7) and 5 (toxicity score = 2.5) in this workshop may only require “visual inspection” as the sole acceptance criterion for cleaning validation.
Provide a method for evaluating cleaning validation using the process capability index (CPK).
Provide a scoring table for assessing cross-contamination risk potential based on the cleaning process capability index (CPu):

The article argues that when assessing cross-contamination risks, the direct severity*probability approach is inappropriate. It introduces the concept of SO (Risk Score) while retaining foundational information.
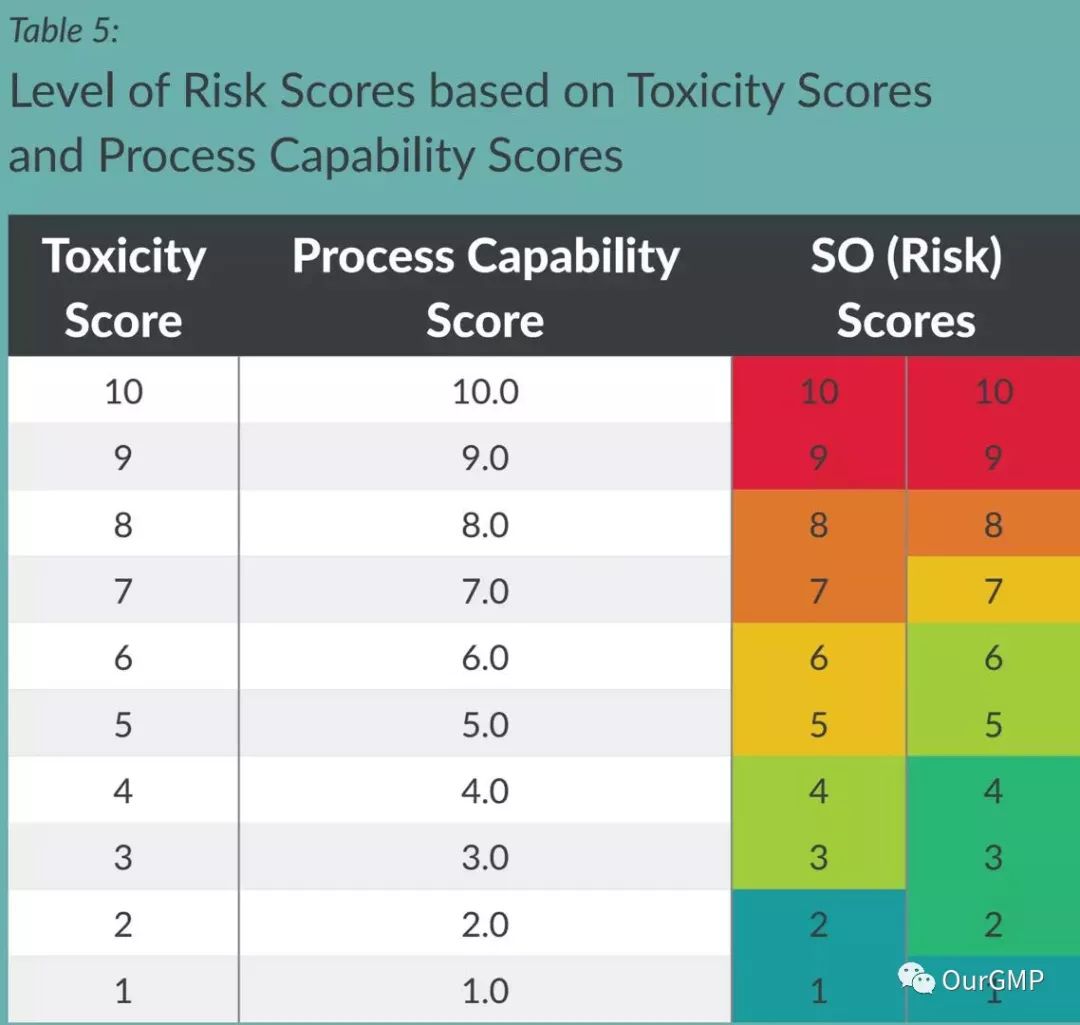
For example, “10/1” indicates a highly toxic compound with very poor cleaning ability, resulting in a highly hazardous situation. “10/1” signifies a highly toxic compound but with highly effective cleaning, leading to a very low-risk scenario. “5/5” indicates a moderately toxic compound with excellent cleaning efficacy, resulting in low risk. “5/10” signifies a moderately toxic mixture with poor cleaning efficacy, posing high risk. “5/1” denotes a moderately toxic compound with highly effective cleaning, thus presenting very low risk.
The article provides justification for employing TOC methods to detect residues in cleaning validation and outlines analytical method validation requirements.

The article notes that regulators have recently relaxed their stance on visual inspection as the sole acceptable standard for cleaning validation, and have provided methods for confirming the detection limits of visual inspections.

The article provides scoring criteria for assessing the severity, likelihood, and detectability of cross-contamination FMEA risks.
Based on the risk assessment results, different risk control measures are proposed for various combinations of toxicity and CPu scores. For instance, a toxicity score of 10 and a CPu score of 10 is unacceptable, necessitating dedicated facilities or single-use equipment for production. Conversely, a toxicity score of 1 and a CPu score of 1 requires only visual inspection after routine cleaning.













