Pharmaceutical 4.0 is an umbrella term encompassing a broad range of technologies and methodologies, including equipment and connected sensors that constitute big data, the Internet of Things, and advanced analytics, as well as new digital biopharmaceutical manufacturing techniques. All these advancements hold potential applications in Maintenance 4.0. However, Maintenance 4.0 requires planning, organization, and execution; it will not materialize spontaneously. This article outlines how to guide maintenance departments and their stakeholders toward Maintenance 4.0.
In the pharmaceutical industry, as in all manufacturing sectors today, safe operations are the paramount prerequisite. However, “operational authorization” is the second essential requirement, and maintenance serves as one of the critical foundational pillars for this authorization. Once equipment is approved for GMP use, maintenance ensures ongoing compliance between production activities or batch sequences and keeps asset performance within the limits of original design standards.
The value of maintenance stems from applied knowledge derived from organized data insights. Modern maintenance organizations blend preventive, predictive, and a degree of corrective maintenance, typically risk-based. Maintenance 4.0 represents a reverse flow output: value-added data is transformed into actionable insights that enable effective maintenance decisions for efficient business operations.
The Maintenance 4.0 transformation requires combining traditional improvements with new tools and technologies, and demands collaborative efforts. Equipping maintenance teams with virtual/augmented reality technologies without a solid foundation—such as robust procedures for planning and scheduling maintenance activities—will not yield true benefits.
Who is responsible for maintaining 4.0?
Without overlooking the importance of enabling technologies, senior management should consider the fact that change management is an indispensable element for sustaining Industry 4.0 applications. From a practical standpoint, internal change advocates and early adopters should play a crucial role in securing plant-level support, communicating rapid successes, and engaging key stakeholders across the entire organization.
Maintenance 4.0 is not a top-down decision but requires the support and engagement of plant-level employees and operators who will ultimately be responsible for its implementation. Maintenance serves several distinct stakeholders, each with their own priorities. These priorities typically fall into operational, commercial, and social categories. The challenge for maintenance managers is balancing social interests (environment, health, and safety), commercial interests (costs as a percentage of total asset value), and operational interests (uptime, overall equipment effectiveness).
Maintenance forms the foundation of a systems lifecycle approach and is highly technical in function. Within the chain of events in a plant's systems lifecycle, maintenance serves as the connecting point between operational activities or batch sequences. Maintenance responsibilities require dedicated department managers who report independently, not to production leadership. The pressure to complete predefined schedules and maintain compliance by executing maintenance tasks during non-related production runs easily leads to prioritizing production. In such cases, maintenance and production leaders should ideally hold equal authority and have the ability to escalate when necessary. Alternatively, establishing a combined schedule that fulfills commercial requirements while allowing for predefined maintenance tasks essential to full compliance provides an integrated approach to system lifecycle management.
The 5 Core Aspects of Maintenance 4.0
Maintenance 4.0 comprises five types of activities designed to keep each asset in a reliable, compliant, and operational state: periodic inspections, prescriptive maintenance, preventive (routine) maintenance, predictive (reliability-centered) maintenance, and corrective/reactive (failure-based) maintenance. These definitions establish a blueprint and framework for a complete, comprehensive, and compliant maintenance function within highly regulated environments.
1. Regular inspections
Inspections provide essential data for understanding equipment status, enabling informed decisions. Optimizing the interval between maintenance inspections minimizes costs associated with both preventive inspections and reactive maintenance. Inspection frequency should be set based on the failure developing period (FDP) by estimating the FDP and establishing an inspection frequency of twice per FDP. For example, if the estimated FDP is four months, the initial inspection frequency would be set to two months. Precise historical factory data serves as the optimal indicator for FDP.
2. Prescriptive Maintenance
Prescriptive maintenance represents the latest evolution in routine maintenance procedures. Unlike predictive practices that merely forecast failures, prescriptive maintenance “prescribes” outcome-focused actions for maintenance and operations based on prescriptive analytics. It is worth noting that this type of maintenance remains in its infancy but is poised to become the next best practice in reliability and maintenance. Prescriptive maintenance enables the most efficient approach to servicing assets, maximizing uptime.
3. Preventive (Routine) Maintenance
Preventive maintenance involves taking proactive measures to prevent unplanned downtime and other avoidable failures. It helps avert unexpected breakdowns on production lines.
4. Predictive (Reliability) Maintenance
Predictive maintenance is a condition-based process that forecasts when equipment failures may occur and provides early warnings. It is achieved by monitoring equipment condition and performing maintenance before anticipated failures. Examples of equipment monitoring methods include: vibration analysis, temperature/infrared thermography, noise/acoustic analysis/listening, visual inspection, fluid analysis (oil), differential pressure, and motion sensors.
5. Corrective/Reactive Maintenance
Corrective and reactive maintenance are performed to ensure equipment can perform its intended functions. Corrective maintenance is executed after a fault is detected, while reactive maintenance occurs after a failure has already happened. It is recommended to use this type of maintenance only on equipment that does not impact product quality.
The maintenance process depicted below represents a comprehensive and inclusive framework for managing all maintenance functions. The gradual maturation of Maintenance 4.0 does not necessitate discarding existing maintenance processes and technologies but rather adapting current practices. Your organization has likely already implemented certain so-called Maintenance 4.0 practices.
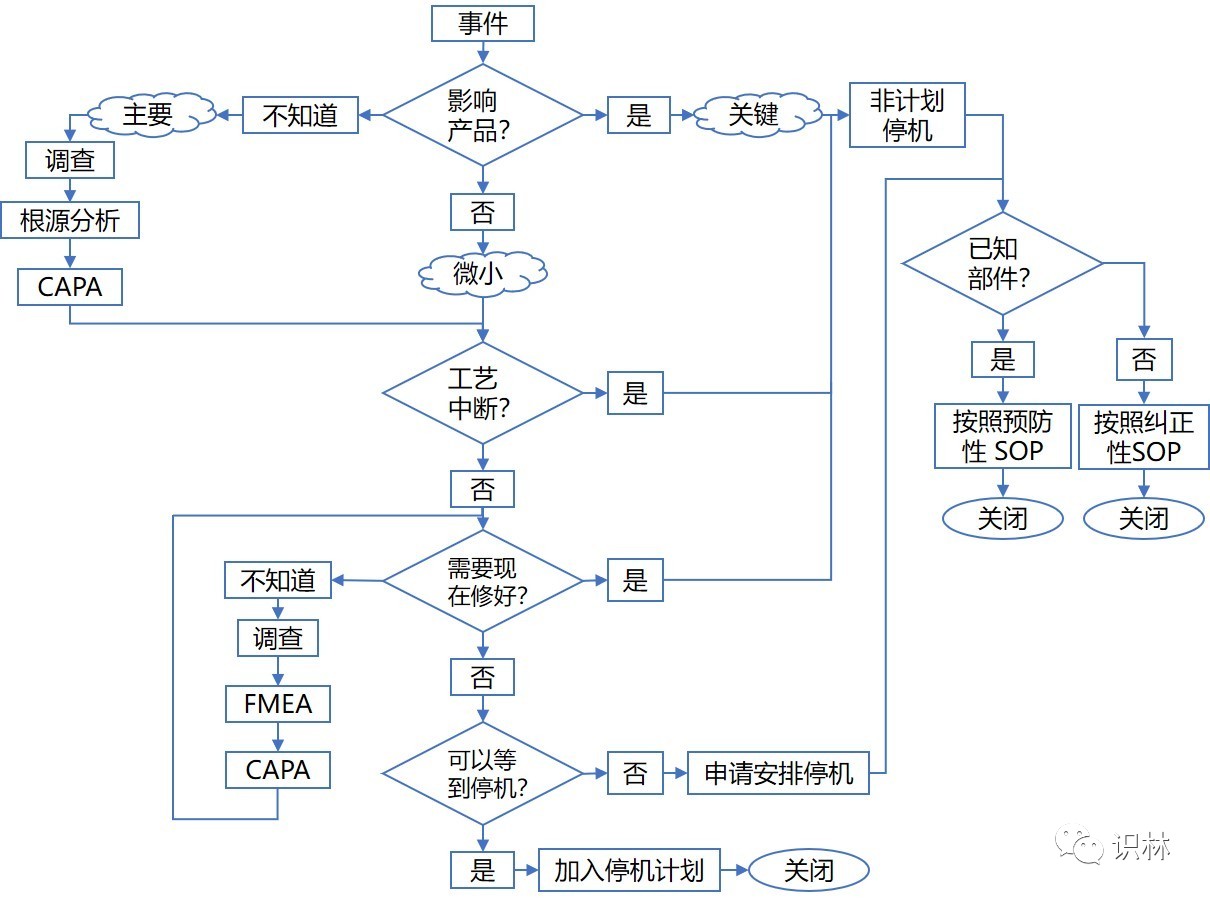
Figure 1: Comprehensive maintenance process comprising four procedures:
(1) Overall Process, (2) Preventive SOP, (3) Corrective SOP, and (4) Shutdown SOP.
Preventive maintenance is time-based, while predictive maintenance is condition-based. Both require work orders approved by the quality department to halt equipment operation, perform repairs, verify post-repair functionality, and return equipment to the operations department. For component failures described in preventive/predictive maintenance work instructions, corrective and reactive maintenance procedures may be covered within the preventive/predictive maintenance program. For component failures not described in these procedures, a work plan must be drafted and submitted for Quality Department approval before maintenance can be performed.
Conclusion
Traditionally, maintenance focused on fixing broken items. Starting with repairing damage (corrective), maintenance has evolved to replacing components before failure (preventive), measuring specific performance indicators signaling potential future failures (predictive), and replacing components proactively. The next phase of maintenance management (Maintenance 4.0) extends beyond prediction to the initial asset stage—commissioning assets and deploying the same tools and techniques to establish design specification metrics from the outset, using indicative metrics to keep assets compliant with their original design specifications.
Proofread by: Shilin-Jiao
References
[1] Maintenance 4.0 – The Next Revolution In Bio/Pharma Manufacturing. Pharmaceutical Online.



