In recent years, isolation technology has rapidly become the preferred choice for personnel operating environments in the production and testing of various types of pharmaceutical products. The operational characteristics of isolators make them highly suitable for the preparation of sterile products and personnel protection. Other applications include the preparation of clinical products, sterile bulk drugs, biologics, sterile cytotoxic products, and sterile testing.
However, this technology lacks authoritative implementation and validation guidelines, and considerable confusion exists between “ISOLATOR” and “RABS.” PDA TECHNICAL REPORT NO. 34 has described ISOLATORS. The healthcare industry lacks a unified definition for isolation technology. The committee recognizes that to provide genuinely useful technical information, reader confusion regarding this technology must be avoided. In some regions, processing environments termed “ISOLATORS” are widely used in clinical pharmacy settings. While these devices share some general characteristics with isolators, they differ significantly from the isolation systems currently employed in the healthcare products industry for sterile product manufacturing and testing.
The PDA recommends the following ISOLATOR definition for the global healthcare industry: An isolator is a sealed enclosure, or one that provides air through a microbiologically filtered system (HEPA Minimum), capable of repeated purification. When closed, material transfer occurs only through purified (where necessary) interfaces or rapid transfer ports (RTPs). When open, materials may enter or exit through designated openings designed and validated to prevent contamination transfer. It may be used for aseptic processing activities, containment of potent compounds, or both aseptic and containment purposes simultaneously.
The PDA proposes the following definition for “RABS”: A barrier system is an open system capable of exchanging contaminants with the surrounding area and cannot be purified to the extent possible within an isolator.
Whether operated in closed or open modes, isolators offer significant advantages over barrier systems: isolators can be purified using repeatable and validated methods, prevent airborne contaminants from entering from the surrounding environment, and block personnel-borne contaminants from entering the isolator. In contrast, barrier systems are open systems that exchange unfiltered air with the surrounding environment, can only be manually sanitized, and allow direct access by personnel wearing gowns.
The increasing use of isolators in the healthcare products industry is particularly notable. In fact, isolators now clearly represent the fastest-growing technology for manufacturing healthcare products. Despite rapid development in this sector, technical guidance for those working in this field remains scarce.
In the early days of the “isoler era,” it was often claimed that isolators were merely scaled-down cleanrooms. Experience has taught scientists and engineers that isolator functional requirements actually differ fundamentally from those of human-scale or “traditional” cleanrooms. While human cleanrooms and isolators share many similarities, they also exhibit significant distinctions.
Isolator Classification: 1. Closed Isolators and 2. Open Isolators
Closed Isolators are subdivided into:
1.1 Sterile-type Closed Isolators
1.2 Safety-type Closed Isolators
1.3 Sterile-Safety Closed Isolators
Open Isolators are subdivided into:
2.1 Sterile-type Open Isolators
2.2 Sterile-Safety Open Isolators
Enclosed Isolators:
Operating as a closed system, these isolators do not exchange unfiltered air or contaminants with the adjacent environment. Their capability to function without personnel entering critical zones enables isolation devices to achieve separation levels between internal and external environments unattainable by other technologies. Due to this effective separation, enclosed isolators are highly suitable for preparing sterile and/or toxic materials.
Three types of closed isolators exist: sterile isolators, safety isolators, and sterile safety isolators. Sterile isolators are designed to exclude external contamination from the critical zone within the isolator. Safety isolators are designed to prevent the release of toxic substances processed within the isolator into the surrounding environment where personnel are present. Sterile safety isolators are designed to prevent the release of sterile, highly active toxic substances processed within the isolator into the surrounding environment where personnel are present.
1.1 Closed Isolators for Sterile Applications
These devices typically operate under positive pressure and undergo verifiable cleaning procedures prior to use. Isolators handling sterile materials adhere to the following general principles:
> They must not exchange air with the surrounding environment unless air passes through microbial retention filters.
> They must be cleaned in a repeatable and quantifiable manner.
> All work or material transfer within the isolator chamber must be performed externally; no person or part of a person may enter the isolator directly during operation.
> All materials entering the isolator must be purified or sterilized and must enter directly through a purification or sterilization system or a rapid transfer port.
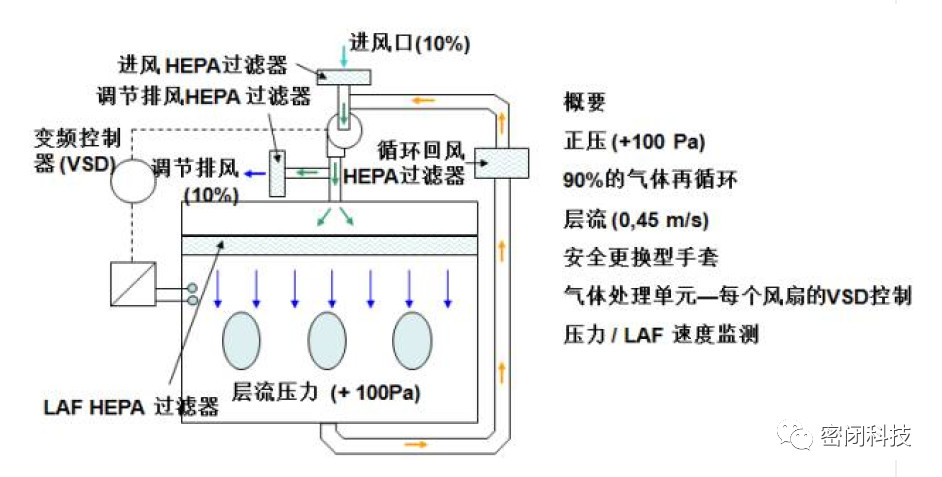

Figure 1: Typical Operating State of a Sterile Isolator
1.2 Enclosed Isolators for Safety Applications
These devices typically operate under negative pressure, purging all potentially hazardous substances before opening. Isolators for safety applications adhere to the following general principles:
> They must not exchange air with the surrounding environment unless air passes through filters capable of protecting the material.
> All work or material handling within the isolator chamber must be performed externally; no person or part of a person may enter the isolator directly during operation.
> All materials leaving the isolator must be clean or transferred in a manner that prevents release of hazardous materials into the surrounding environment.
> Purification must be performed in a repeatable and quantifiable manner.
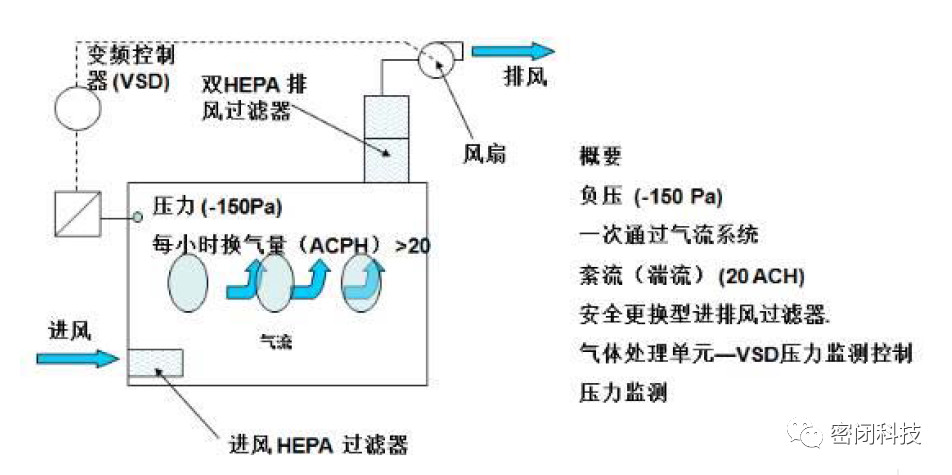

Figure 2: Typical Operating State of a Safety Isolator
1.3 Enclosed Isolation Devices for Sterility and Protection
These devices are constrained by the two isolator types described above. They typically operate under positive pressure and incorporate additional safety measures such as negative pressure airlocks. Workers may consider using supplemental personal protective equipment.
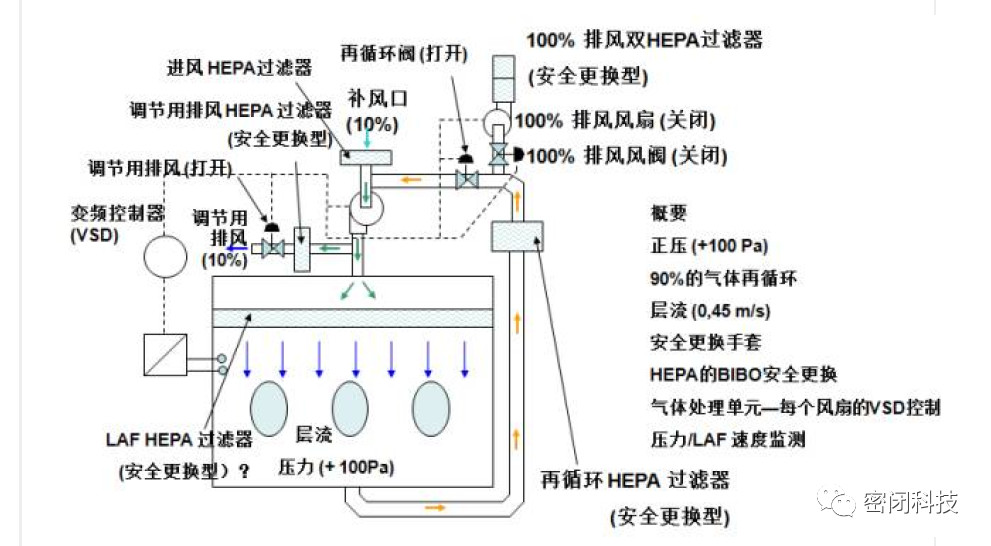

Figure 3: Typical Operating State of a Sterile Protection Isolator

Open-type isolator
开放式隔离器与封闭式隔离器的不同之处在于,开放式隔离器的设计允许在运行期间连续或半连续地进出物料,同时保持对内部环境的保护水平。开放式隔离器不与周围环境交换未过滤的空气或污染物。打开的隔离装置在关闭时被净化,然后在生产过程中打开。开放式隔离器通常用于成品药品的无菌灌装。
2.1用于无菌应用的开放式隔离器
除了在生产过程中存在接口外,开放式隔离器满足与上述封闭式隔离器相同的一般原则。有许多设计可以成功地管理灌装容器组件的进出,同时防止污染从周围环境进入。
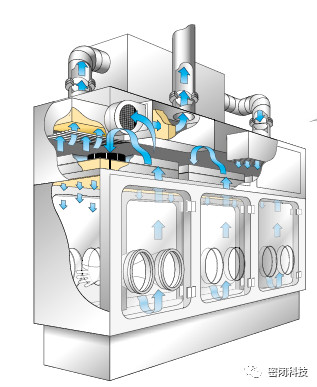

Figure 4: Open Isolator for Sterile Applications
2.2 Open Isolators for Sterile and Protective Applications
These isolators resemble standard open isolators and safety-enclosed isolators. For these systems, the design typically incorporates a cleaning process to remove any toxic contaminants from the exterior of sealed containers before discharge from the isolator outlet. The design must also prevent air ingress to maintain the sterile nature of the internal environment.

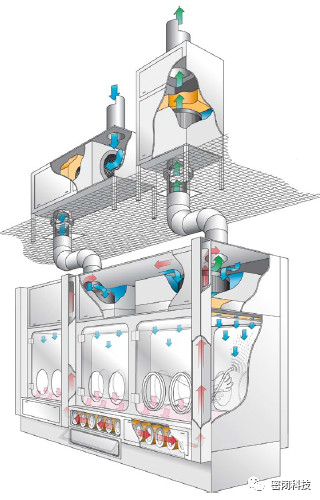
Figure 5: Open Isolator for Sterile High-Toxicity Applications
The diagram below outlines some operational characteristics and applications of isolators, barrier systems, and cleanrooms. Note that the system at the edge of the drawing is essentially equivalent, providing minimal protection for materials and personnel.
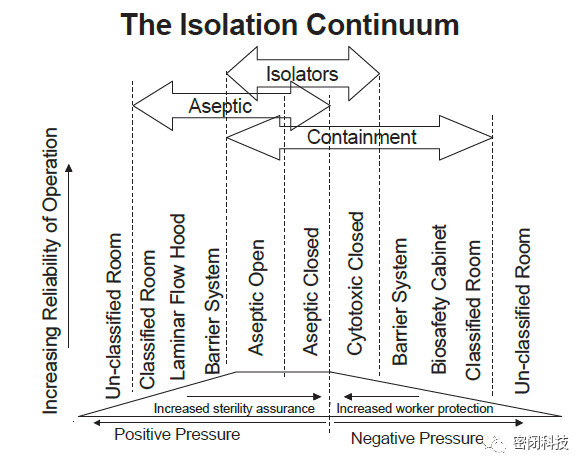

Figure 6: Isolator Barrier System and Cleanroom Applications
Copyright Notice:
Copyright belongs to Aoxing. Authored by Race Li, sourced from the WeChat public account “Sealed Technology.”
For reprinting, please clearly indicate the author and source of the article.








