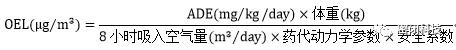Original: Jordan Song
Pharmaceutical products are increasingly developing toward high toxicity and high potency, with exposure limits becoming progressively stricter. Since drugs can be absorbed through skin and mucous membranes, any potential skin contact requires special attention. This is why the containment performance of equipment has become critically important.
The evaluation of containment performance addresses three key aspects:
The potential for pharmaceuticals to harm worker health
The potential for pharmaceutical cross-contamination
The potential for pharmaceutical environmental pollution
Conducting containment performance testing serves multiple purposes:
Identifying equipment issues prior to shipment
Providing data for future project development
Meeting customer expectations
Certification by registered industrial hygienists
Enhancing equipment competitiveness in the market
Ensuring employee safety
In 2014, the EU GMP published updates on managing cross-contamination risks. The new GMP requires risk assessments for products manufactured in shared facilities and further specifies that independent facilities should be used in the following situations:
1. Inadequate risk control
2. Scientific data from toxicological evaluations do not support risk manageability
3. Residue limits cannot be reliably detected using validated analytical methods
ADE: Acceptable Daily Exposure 每日可接受剂量
OEL:Occupational Exposure Limit 职业暴露限值
OEB=Occupational Exposure Band 职业暴露带
The new GMP requires that in multi-product facilities, a risk assessment for drug cross-contamination must be conducted. As required, the ADE of the drug must be determined. Based on the ADE, the OEL can be calculated.
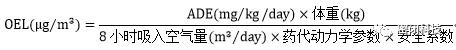
OEB is a classification system based on OEL levels, with different pharmaceutical companies or institutions employing varying classification methods. The five-level classification is one of the most commonly used approaches:
Design of Detection Method
ISPE Good Practice Guide: ‘Assessing the Particulate Containment Performance of Pharmaceutical Equipment’
Prepared by the SMEPAC Committee (Standardized Measurement of Equipment Particulate Airborne Concentration)
This guide defines relevant operational specifications and provides benchmarks and improvement methods for organizational practices. It offers a methodology for handling pharmaceutical-related materials, facilitating the assessment of potential risks.
1. Set target value: CPT (Containment Performance Target), which should generally be less than or equal to the drug's OEL value.
4. Test personnel: Equipped with protective gear to prevent contamination introduction
5. Sampling methods: Operational space sampling, personnel respiratory sampling, surface wipe sampling, etc.

4. Test personnel: Equipped with protective gear to prevent contamination introduction
5. Sampling methods: Operational space sampling, personnel respiratory sampling, surface wipe sampling, etc.

6. Sampling Point Layout: Select sampling points, sampling times, and frequencies based on relevant principles.
7. Test Samples and Issue Reports