Original: Mei Zhe, Xia Rui
Pharmaceutical Affairs Review
USP40<1174> POWDER FLOW
Numerous methods exist to characterize the flow properties of powders widely used in the pharmaceutical industry. Many pharmaceutical literature attempts to correlate various powder flow measurement techniques with production characteristics. The multifaceted nature of powder behavior complicates the study of powder flow.
The purpose of this chapter is to review the most common methods for characterizing powder flowability in the pharmaceutical literature. No single simple test method can fully characterize the flowability of pharmaceutical powders. This chapter proposes standardized powder flowability test methods that are valuable during drug development.

Four commonly used methods for characterizing powder flowability are:
(1) Angle of repose
(2) Compressibility coefficient or Hausner ratio
(3) Pore flow velocity
(4) Shear cell method
Given the number of testing methods and variables, adopting standardized testing methods whenever possible is advantageous. With this goal in mind, the most commonly used methods are discussed below, along with important experimental considerations and recommendations for method standardization. Generally, any method measuring powder flowability should be practical, useful, reproducible, sensitive, and yield meaningful results. No single method can fully and completely characterize a powder's flow properties. One strategy is to employ multiple standardized test methods to characterize different aspects of powder flowability relevant to pharmaceutical scientists.
Angle of Repose
The angle of repose is a characteristic of friction or resistance between particles. Reports indicate that angle of repose results are highly dependent on the method employed. A challenge with this test arises when powder agglomerates or forms voids upon cone formation, leading to material stratification. Despite these difficulties, the method remains widely used in the pharmaceutical industry and has demonstrated value in predicting production issues in the literature.
The angle of repose is a constant, and several different determination methods assume the cone formed by the material represents a three-dimensional angle (relative to the base) (briefly described below).
Basic Methods for Angle of Repose
The literature describes multiple angle of repose testing methods. The most commonly used methods for determining the static angle of repose can be categorized based on two important experimental variables:
(1) The height of the “funnel” through which the powder passes relative to the base can be fixed or can vary as the cone forms.
(2) The diameter of the cone's base can be fixed or can change as the cone forms.
Relationship Between Angle of Repose and Flowability
Although using the angle of repose to characterize powder flowability involves some uncertainty, most pharmaceutical literature aligns with Carr's classification, as shown in Table 1. Examples in the literature indicate that an angle of repose between 40° and 50° can meet production requirements, while values exceeding 50° are rarely acceptable for manufacturing purposes.
Table 1. Correspondence Table of Fluidity and Angle of Repose
Fluidity
|
Fluidity
|
Angle of Repose (°)
|
|
Excellent
|
25-30
|
|
Good
|
31-35
|
|
Fair
|
36-40
|
|
Acceptable
|
41-45
|
|
Poor
|
46-55
|
|
Very Poor
|
56-65
|
|
Extremely Poor
|
>66
|
Experimental Recommendations for the Angle of Repose
The angle of repose is not an inherent property of powder materials; that is, it is highly dependent on the method used to form the cone. The following important recommendations have been proposed in the existing literature:
-
The peak of the powder cone may deform due to the influence of the upper powder layer. By carefully constructing the powder cone, deformation caused by impact can be minimized.
-
The nature of the cone's base influences the angle of repose. It is recommended to form powder cones on a “common base,” which can be achieved by forming cones on the powder layer. This can be accomplished using a fixed diameter and a protruding outer edge, thereby retaining a layer of powder that forms the cone.
Recommended Method for Determining the Angle of Repose
It is recommended to determine the angle of repose on a fixed base retaining a layer of powder. The base should be vibration-free. Adjust the height of the funnel to carefully form a symmetrical cone. When moving the funnel, take care to prevent vibrations. During cone formation, maintain the funnel height approximately 2-4 cm from the top to minimize powder impact on the cone tip.
If a symmetrical cone cannot be successfully formed or is not reproducible, this method is unsuitable. In addition to the above pouring method, alternative techniques include the discharge method and dynamic angle of repose determination.
Determining the angle of repose involves measuring the height of the cone and the base, then calculating the angle of repose α.
The calculation formula is as follows:
tan(α) = height / radius
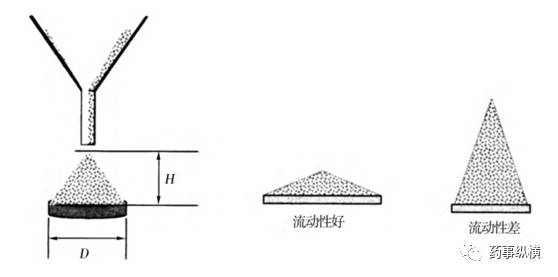
Principle of Angle of Repose Measurement
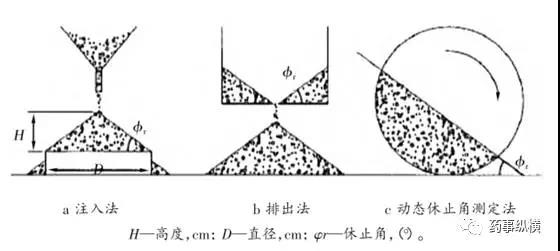
Classification of Angle of Repose Measurement Methods
Compression ratio and Hausner ratio
In recent years, the compression coefficient and its closely related Hausner ratio have become simple, rapid, and popular methods for predicting powder properties. Since a material's bulk density, size and shape, surface area, moisture content, and viscosity influence the compression coefficient, it has been proposed as an indirect indicator for characterizing powder flowability.
The compression coefficient and Hausner ratio are determined by measuring the powder's tap density and bulk density.
Determination Methods for Compressibility Coefficient and Hausner Ratio
The methods for determining the compression coefficient and Hausner ratio exhibit some differences, but the fundamental approach involves measuring (1) the uncompacted apparent volume V0 and (2) the compacted apparent volume Vf.
The formulas for calculating the compression coefficient and Hausner ratio are as follows:
Compression coefficient (%) = 100 × [(V₀ − V_f)/V₀]
Hausner ratio = (V₀/V_f)
The compression coefficient and Hausner ratio can also be calculated using bulk density (ρbulk) and tapped density (ρtapped):
Compression coefficient (%) = 100 × [(tapped density − bulk density) / tapped density]
Hausner ratio = (tapped density / bulk density)
For the compression coefficient and Hausner ratio, the acceptable ranges under different flow states are shown in Table 2.
Table 2. Compressibility Factor and Hausner Ratio under Different Flow Conditions
|
Fluidity
|
Compressibility Index (%
|
Hausner ratio
|
|
Excellent
|
≤10
|
1.00-1.11
|
|
Good
|
11-15
|
1.12-1.18
|
|
Fair
|
16-20
|
1.19-1.25
|
|
Acceptable
|
21-25
|
1.26-1.34
|
|
Poor
|
26-31
|
1.35-1.45
|
|
Very Poor
|
32-37
|
1.46-1.59
|
|
Extremely Poor
|
>38
|
>1.60
|
Experimental Considerations on Compressibility Factor and Hausner Ratio
The compression coefficient and Hausner ratio are not intrinsic properties of powders; they are highly dependent on the testing method employed.
The following critical factors influence the determination of the uncompacted apparent volume V0, compacted apparent volume Vf, bulk density ρbulk, and tapped density ρtapped:
- Diameter of the measuring cylinder used
- Number of taps applied to the powder to achieve the tapped density
- Mass of the material used in the test
- Rotational speed of the sample during compaction
Recommended Measurement Methods for Compressibility Coefficient and Hausner Ratio
Use 100 g of test sample with a 250 mL graduated cylinder. A smaller sample and graduated cylinder volume may be used, but the method parameters should be described. Three parallel determinations are recommended.
The flow rate of a substance depends on numerous factors, some particle-related and others process-related. Monitoring the bulk flow rate of a material is a reliable method for determining powder flowability. Continuous monitoring of flow performance is particularly crucial, as pulsed flow patterns have been observed even in free-flowing materials. Changes in flow rate during container emptying can also be observed. Flow rate correlates with pore size, particle size, and particle density. Bulk flow rate is applicable only to freely flowing substances.
Pore flow rate is the volume of material discharged from a container (measuring cylinder, funnel, hopper) per unit time. Flow measurement can be either intermittent or continuous.
Fundamental Methods for Determining Pore Flow Velocity
The most common methods for determining pore flow velocity are classified based on the following three key experimental variables:
(1) Type of container used to hold the powder. Common containers include graduated cylinders, funnels, and hoppers in production equipment.
(2) Size and shape of the discharge orifice. Orifice dimensions and geometry are critical factors in determining powder flow velocity.
(3) The method used to measure powder flow rate. Flow can be measured continuously using an electronic balance equipped with a recording device (strip chart recorder, computer), or measured in discrete samples (non-continuous measurement).
Variables in Pore Flow Velocity Measurement Methods
Both mass flow and volumetric flow can be measured. Mass flow measurement is relatively straightforward but is more suitable for high-density materials. Since mold filling involves a specific volume, measuring volumetric flow is the optimal choice.
Sometimes it is necessary to connect a vibrator to facilitate material discharge from the container, but this complicates the interpretation of results. A movable orifice device has been proposed to more closely simulate rotational pressure conditions. The minimum diameter of the orifice through which powder flows can also be determined.
Common scales for pore flow velocity
There is no universal scale, as flow rates are highly dependent on the measurement method used, making comparisons with published results difficult.
Experimental Recommendations for Pore Flow Velocity
Pore flow velocity is not an inherent property of powders but is largely dependent on the measurement method employed. Several key factors influencing these methods have been discussed in the literature:
-
Orifice diameter and shape.
-
Container material type (metal, glass, plastic).
-
Powder bed diameter and height.
Recommended Method for Pore Flow Velocity
Pore flow velocity is only applicable to materials with a certain degree of fluidity and is unsuitable for viscous substances. A cylinder may be used as the container, as it has minimal impact on flow rate.
The flow rate of this apparatus is determined by the movement of the powder rather than its displacement along the container wall. Powder flow often increases when the height of the powder column is less than twice its diameter. The orifice should be circular, and the cylinder should be vibration-free.
General specifications for column dimensions are as follows:
Orifice diameter > 6 times the particle diameter
Cylinder diameter > 2 times the orifice diameter
Using a hopper as a container may be appropriate as it represents the flow characteristics under production conditions. Using a funnel is not advisable, especially one with a long neck, since the flow rate will be determined by the size and length of the funnel opening, as well as the friction between the long neck and the powder. A truncated cone may be suitable, but the flow rate will be affected by the friction coefficient of the powder walls, making material selection a critical consideration.
For the cylinder opening, a flat plate should be used, with different orifice plate diameters available to provide maximum flexibility and better ensure the powder flow pattern. Flow measurement can be intermittent or continuous. Continuous measurement using an electronic balance allows for more effective monitoring of instantaneous flow changes.
To establish powder flowability research and hopper design on a more fundamental basis, various powder shear testers and more thorough and accurate methods for determining powder flow properties have been developed. The shear cell method has been widely adopted in pharmaceutical material research.
In this method, various useful parameters can be obtained, including the yield trajectory representing the shear stress-shear strain relationship, the angle of internal friction, the unconfined yield strength, the tensile strength, and various derived parameters such as the flow factor and other flow indices. Due to the ability to more precisely control experimental parameters, flowability can also be determined as a function of consolidation time and other environmental conditions. This method has been successfully applied to determine the critical parameters of hoppers.
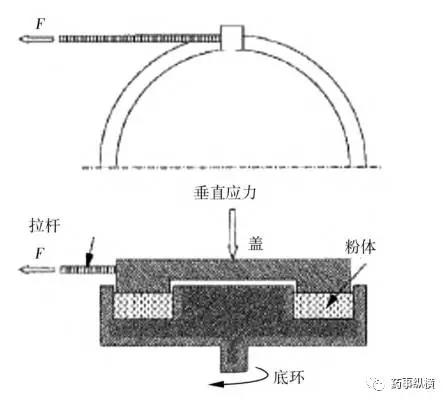
Basic Methods of the Clip Pool
One type of shear cell is the horizontal shear cylindrical shear cell, which creates a shear plane between a lower fixed base and the movable section of the shear cell ring.
The annular shear cell design offers certain advantages over the cylindrical shear cell, including reduced material requirements. However, a drawback is that it results in non-uniform shearing of the powder bed, meaning more material is present on the outer side of the ring than on the inner side. The third type of shear cell (plate-type) features a thin layer of powder between a lower fixed rough surface and a movable upper rough surface.
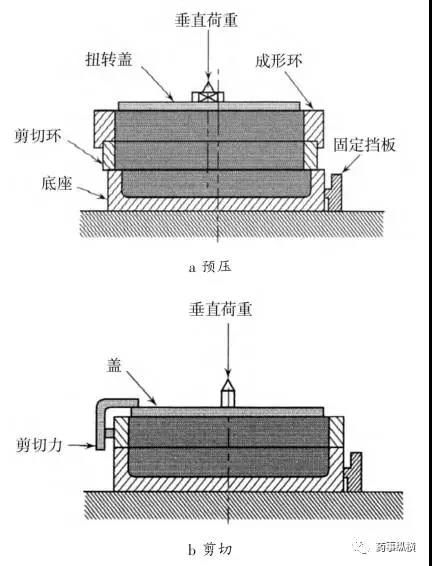
Schematic Diagram of the Jenike Shear Test Apparatus
Schematic Diagram of the Jenike Test Method
Ring Shear Test Method
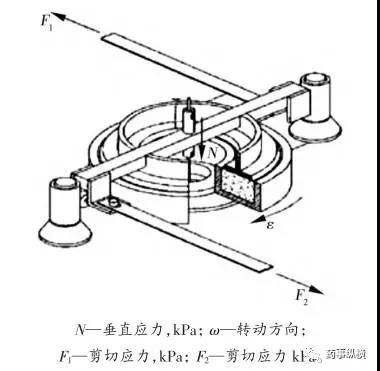
Schematic Diagram of the Schulze Shear Apparatus
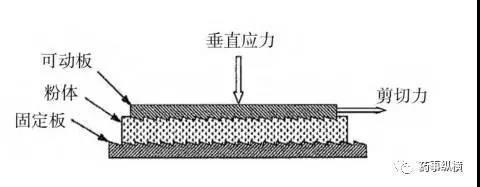
Parallel Plate Shear Testing Apparatus Diagram
All shear cell methods have their advantages and disadvantages, which are not discussed in detail in this chapter. As with other methods used to characterize powder flowability, numerous variables are described in the literature.
Generally, a significant advantage of the shear cell method is the greater degree of experimental control it offers. However, this method is highly time-consuming and requires substantial material quantities and experienced operators.
Recommendations for the Shear Pool Method
Many existing shear cell devices and test methods provide a wealth of data that can be effectively utilized to characterize powder flowability. They also aid in the design of equipment such as hoppers and silos.
Given the diversity of available equipment and experimental procedures, no specific recommendations regarding usage methods are provided in this chapter. When employing the shear cell method to describe powder flow characteristics, it is recommended to include a complete description of both the equipment and methodology.
(1) Measuring the angle of repose is the most commonly used method for assessing powder flowability in solid dosage form development. Many formulation developers obtain a general understanding of powder flowability through angle of repose measurements. However, this method has the following drawbacks:
① Measurements are taken under pressure-free conditions, failing to represent actual production conditions.
② It reflects interparticle interactions rather than revealing the intrinsic properties of the particles themselves.
③ Measurement results depend on the testing equipment and conditions, exhibiting poor reproducibility.
(2) The compression coefficient and Hausner ratio offer the advantages of simple calculation and rapid comparison of differences in active pharmaceutical ingredients, excipients, and formulation flowability. They hold significant application value in capsule production, but also present drawbacks:
① The compression coefficient and Hausner ratio cannot accurately reflect the actual flowability of different powder materials.
② Although USP<616> includes standardized methods for determining bulk density and tap density, the lack of external pressure control introduces randomness into the vibration compaction process.
(3) The pore flow velocity method has the following drawbacks:
① Flow velocity measurements are highly influenced by experimental conditions such as pore diameter; different pore sizes yield different flow velocities.
② It cannot measure powders with strong interparticle adsorption forces that prevent flow.
(4) Among methods for measuring powder flowability, the shear cell method offers quantifiable and reproducible results, but its drawbacks include:
① Requiring a relatively large amount of powder for testing.
② Taking a considerable amount of time to perform.
③ Requires specialized equipment.
Nevertheless, the shear method is grounded in more fundamental theory and provides more reliable results than the simpler methods described earlier.
During solid dosage form development, powders that flow smoothly in laboratory-scale trials often encounter flow issues during pilot-scale upscaling and production-scale manufacturing. This leads to inconsistent product quality and, in severe cases, production stoppages. In pharmaceutical manufacturing, powder transfer—such as discharge from funnels, mixing and discharge from blenders, and filling into capsules or tableting dies—all depend on powder flow properties. Powder flow fundamentally arises from unbalanced forces acting on particles.
Analysis of particle forces reveals that gravity, interparticle adhesion, friction, and electrostatic forces all act upon particles, with gravity and interparticle adhesion exerting the greatest influence on powder flow. Particle size distribution and shape have been proven to affect powder flowability. Additionally, factors such as temperature, moisture content, electrical properties, bulk density, binding index, and internal friction coefficient are also recognized as influencing the flowability of pharmaceutical powders.
Given the multitude of factors affecting powder flowability, and considering that the flow of pharmaceutical powders significantly impacts mixing efficiency, production rates, and formulation uniformity, employing scientifically sound methods to measure powder flowability and investigate its influencing factors will aid in resolving these challenges.
Researching the fundamentals of powder science to gain a deep understanding of the “structure-property” relationships of materials and their connections to formulations, processes, equipment, and product quality is a critical step in solid dosage form development. Analyzing the application of the shear cell method for measuring pharmaceutical powder flowability will enable researchers to deepen their understanding of the factors influencing powder flowability in the pharmaceutical field and the methods for improving it.











