The purpose of any mixing operation is to ensure uniformity—initially within the mixing chamber of the mixer, though what truly matters is the distribution characteristics of the powder in the retail packaging. In many cases, raw materials are blended in nearly equal proportions (e.g., a 50:50 ratio); however, from a process perspective, it becomes more challenging when trace elements (active ingredients) must be effectively and uniformly mixed with other raw materials present in higher proportions within the formulation. Determining whether these micro-components are optimally distributed involves more than just selecting the right solid mixer; in fact, the size of the raw material particles is often a more critical factor. Product quality heavily depends on the continuous weighing process upstream of the continuous mixer; any inaccuracies occurring during feeding will ultimately manifest in the final product, as the actual formulation will deviate from the desired target values. The uniformity of powder mixing is typically verified through trial measurements. Online measurement using PAT sensors and analyzers enables direct determination of mixing quality within the process. For instance, a near-infrared analyzer can be installed at the outlet of a continuous mixer to measure active ingredient concentration online, thereby assessing mixing uniformity.
The fluctuation range of actual mixing uniformity—such as the variance in active ingredient concentration across a sample set—determines the estimated mixing quality or uniformity. A smaller fluctuation range (correspondingly, lower variance) indicates superior mixing performance.
Assessing mixing uniformity depends on the number of samples tested and the sampling method employed. It is often overlooked that variance (as a measure of mixing quality or uniformity) decreases with increasing sample weight per unit. Even for identical mixtures, a 10 g sample exhibits significantly higher mixing variance than a 1 kg sample.
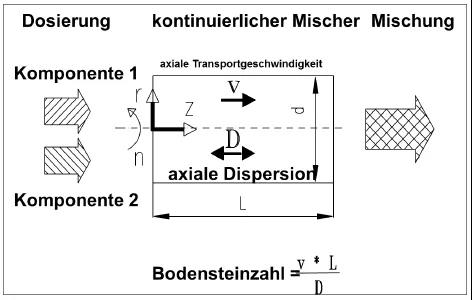
Random particle motion causes varying residence time distributions. For axial mixing, this positively attenuates time-limited feed fluctuations [3]. Mixer vessel and agitator designs ensure effective control of both axial and radial mixing, with residence time distribution also being controlled. Average mixing residence times range from 5 to 50 seconds. The average residence time of particles within the mixing chamber is influenced by several process variables [3]:
● Feed rate of raw materials into the continuous mixer, ranging from 10 to 500 kg/h. Higher flow rates result in shorter average residence times;
● Opening angle of the outlet baffle plate;
● Rotational frequency of the mixer;
● Shape and pitch angle of the mixing blades.
Following Danckwerts' design concept, the efficiency of continuous mixers is described using the variance reduction ratio [3]. A lower concentration variance indicates better mixing uniformity, which is why the variance of mixing uniformity—or the closely related concept of relative standard deviation—is frequently used as an indicator of mixing quality.
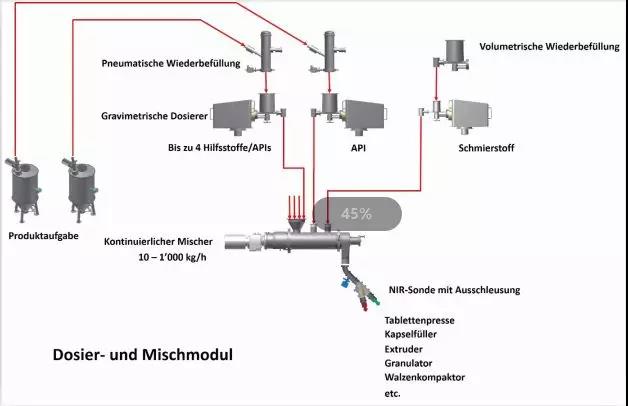
Weight-type feeders generally operate based on the “loss-in-weight” principle. Their purpose is to ensure a constant feed flow rate (mass flow). The unit commonly used for mass flow is kg/s. Feed flow can be described as the change in mass (∆m) over a given time interval (∆t), typically measured in kilograms, which may span seconds or hours. For continuous feeding, high-resolution weighing technology determines how much mass (∆m) the feeder loses (weight loss) during short time intervals (∆t). The rotational speed of the metering screw is regulated by a dedicated control unit. This approach enables the mechatronic system to achieve constant mass flow, incorporating weighing and control technologies. Loss-in-weight feeders require uninterrupted operation over extended periods without production stoppages due to raw material depletion; this is achieved through pneumatic feeding systems. In such cases, raw materials are pneumatically conveyed via fine tubes to vacuum feeders, thereby replenishing the loss-in-weight feeder. Fine active ingredients and excipients demand complex mechanical solutions and control systems. For instance, twin-screw loss-in-weight feeders deliver viscous materials into mixers with high precision and constant speed. Additionally, the pharmaceutical industry imposes stringent requirements for equipment safety and cleanliness. These demands are met through the consistent use of sealed, quick-release fasteners. Alternative solutions can isolate contaminated feed hoppers from gears and weighing devices, enabling their safe cleaning and processing through autoclaves. Loss-in-weight feeders should be integrated with continuous mixers. Safety and ease-of-cleaning requirements must be considered early in the design layout of feeding and mixing modules.
Compared to the large capacities required in other industries, mixers demanded by the pharmaceutical sector are typically considered miniature mixers due to their small volume, ranging from 1 to 15 liters. However, it is precisely this miniaturization that enables the equipment to be implemented in intriguing ways. The suspended mixing rotor serves as an excellent example. This bearing method allows the mixing chamber, rotor, and shaft seal to be separated from the fixed drive in seconds. Consequently, cleaning these micro-machine components requires minimal time, and sterilization is achieved by exposing them to high-pressure steam. The compact design facilitates easier integration of the micro-mixer or entire feed mixing module into a unified system.